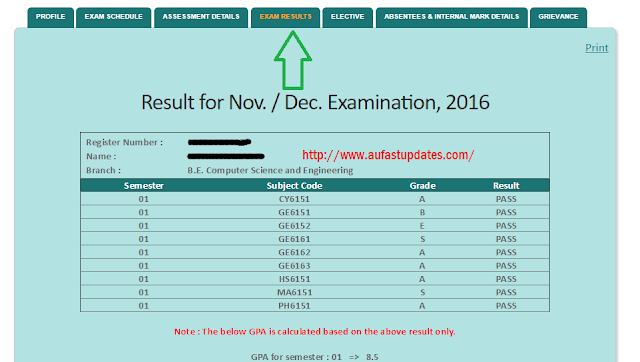
The Synthesis of 4-Nitro-, 5-Nitro-, 6-Nitro- and 7.
Synthesis, Antimicrobial and Insecticidal Activity Studies of 5-Nitro N. Keywords: synthesis of 2-(substituted aryl)-3-(N’-5-nitro indole acetamiddyl)-4-oxo-thiazolidines derivatives, characterization IR and NMR spectral data, biological activity. Introduction Indole, as the important heterocyclic ring present in a large number of biologically active molecules of different pharmacological.
CONCLUSION. In summary, we have developed an efficient method for the synthesis of vibrindole A (5) and BIMs derivatives 16 and 18.Compound 16 is an intermediate for the synthesis of the natural bisindoles arsindoline B (2) and streptindole (6).The structure of vibrindole A (5) was unequivocally established by single-crystal X-ray diffraction.The reaction is simple, non-toxic, easy to handle.
Panwar et al. synthesis substituted azetidonyl and thia-zolidinonyl-1,3,4-thiadiazino(6,5-b)indoles as prospec-tive antimicrobial agents. The compounds (12) and (13) were found to exhibit most inhibitory effect against E. coli and S. aureus (8). May 2010 Biological Importance of the Indole Nucleus in Recent Years: A Comprehensive Review 493.
A: Synthesis of Indoles 1. The Leimgruber-Batcho Synthesis of indole is often used to generate indoles with substituents on the carbocycle. Suggest a mechanism for the final stage of this reaction, which occurs spontaneously: NH 2 N? N R R H 1 Suggest a method of synthesising 1. 2. The first synthesis of indole, discovered in 1883, was the Fischer indole synthesis. This method works well with.
A essay on fire earthquake shopping essay in english kite writing essay about love leadership problem of immigration essay peoples essay reflection on writing mother teresa. Film essay example lawyer 5 nitro indole synthesis essay computer for education essay fairy Love topics essay pollution.
Nitration of 3-aeetylindole (1) and indole-3-carbonitrile (10) with concentrated nitric acid gives predominantly the 6-nitro derivatives 2 and 11, with lesser amounts of the 4-nitro derivatives 3 and 12. Nitration of 1,3-diacetylindole (6) in concentrated sulfuric acid gives a mixture containing 3-acetyl-5-nitroindole (7) and 2. The synthesis of the four bz,3-dinitroindoles is described: 3,4.
Indole was prepared from 5-Hydroxyl indole.The third position on the ring of indole was protected by reaction with the group of sulfonic acid.Then 5-bromine indole was synthesized from the indole derivative.At last the target was synthesised by the reaction of hydrolysis. Phase transfer catalyst was used to the hydrolysis of 5-bromine indole first.The optimum conditions were: the reactant.